How Canada is driving climate change innovation
Communities around the world increasingly feel the impacts of climate change, which is driving a ...

The manufacture of a drug is a very long process that takes an average of 15 years from the initial discovery of the active substance to its commercialization. This process includes many steps such as research and development, clinical trials, evaluation, and approval by health authorities. To market a drug, companies must obtain marketing authorization (MA) for that drug from the relevant agencies (e.g., EMA for Europe, FDA for the United States).
The main mission of these agencies is to authorize and monitor drugs marketed in authorized countries. If companies obtain this authorization, they can then produce the drug on a large scale and market it in authorized countries.
Laws and regulations relating to the marketing of drugs have been gradually put in place over the last few centuries to establish rigorous and mandatory controls to prevent serious accidents. Several steps are required to obtain this authorization.
In general, the company must document the qualities of the drug in an extremely precise and rigorous manner according to three criteria: Quality – Toxicity/Safety – Efficacy.
Scientific evidence must be provided (i.e., role of clinical trials) because commercial success is not a proof of safety and efficacy. A “risk-benefit” analysis (toxicity/efficacy) is then carried out by the regulatory authorities to assess the value of a drug.
You should note that obtaining authorization does not mean the end of controls.
Once the drug is on the market, the companies that hold the authorization are required to continue monitoring the drug’s “behavior” throughout its life cycle. This is called pharmacovigilance, and the data is collected and sent to the relevant health authorities.
Learn how Leyton can help you fund and de-risk your next drug’s path to market, speak with a life sciences funding expert today.
Explore our latest insights
More arrow_forward
Communities around the world increasingly feel the impacts of climate change, which is driving a ...

Harnessing Science for Ecosystem Protection Canada, with its immense ecological diversity, from A...

The Canadian mining sector is transforming. Automation, electrification, and real-time data integ...
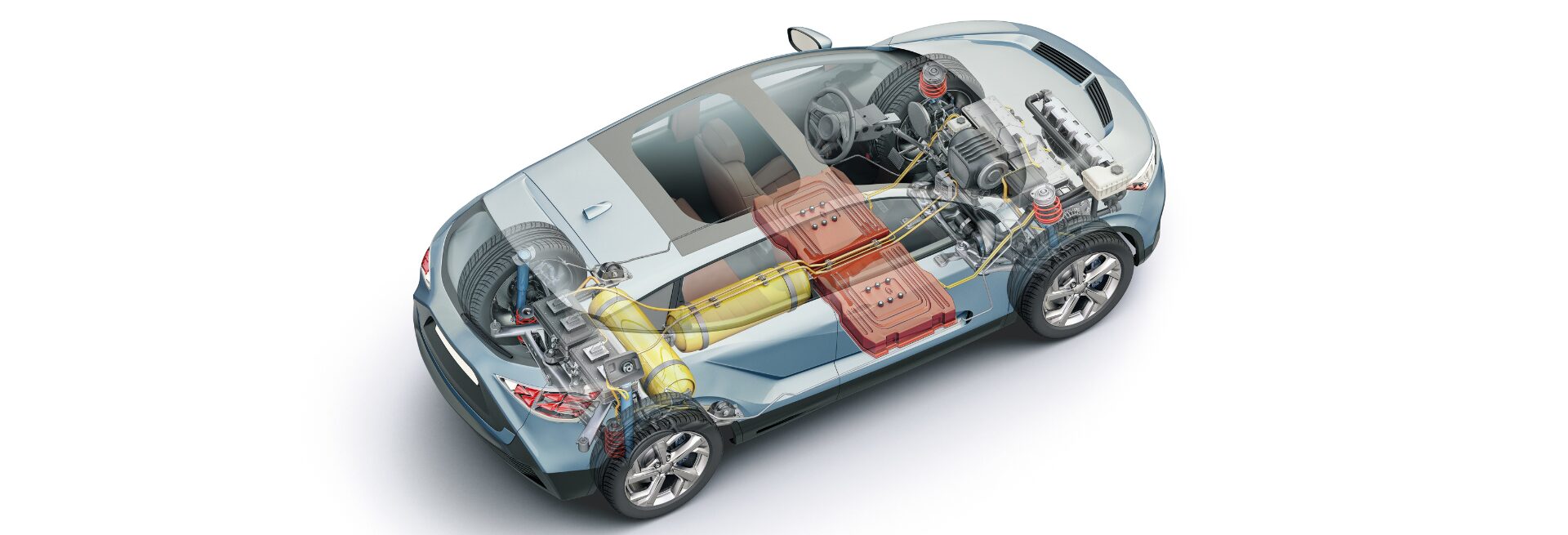
The race toward cleaner, more efficient transportation has accelerated research into lighter, sma...