Tradition & Technology: A New Era of Biodiversity Manageme...
Harnessing Science for Ecosystem Protection Canada, with its immense ecological diversity, from A...

The quiet killers of life science innovation for biotech startups are cash flow, compliance, and product complexity. Many founders often overlook critical factors when excited by innovation, resulting in a failure to recognize the blind spots associated with pursuing any biotechnology venture.
In this article, we will cover the first “C,” cash flow, and explore the often-overlooked processes in the biotech funding space. We will also discuss the mistakes people make in addressing their cash flow using some public press examples and how these can be mitigated.
When a founder comes up with a new idea and finds supporting literature, they often believe that the path to a successful final product is straightforward.
However, this is far from the truth. Many underestimate the time and costs required to reach regulatory approvals or market milestones.
For example, Rubius Therapeutics, a biotech company based in Cambridge, Massachusetts, liquidated its assets just four years after going public at a valuation of $2 billion. This was due to its failure to demonstrate efficacy in trials for red blood cell therapies.
Similarly, IMV Inc., a Halifax-based biotech company specializing in immuno-oncology vaccines, filed for bankruptcy only five years after its IPO. This occurred after clinical delays and challenges in securing partnerships left them without a viable commercial product to support their cash flow.
Development is a long game, so maintaining cash flow is crucial for keeping a company afloat during product failures or shifts in direction. One way to help manage cash flow issues is by filing for the Scientific Research and Experimental Development (SR&ED) program each year, which can cover up to one-third of R&D-related salaries.
This assistance can help delay layoffs and provide some level of cash flow, even when R&D efforts do not yield successful results.
Equity-based fundraising can dilute ownership quickly if SR&ED and other tax offsets aren’t utilized. Achaogen Inc. raised over $300 million in equity-based funding and had developed two anti-infective drugs, approved by the FDA in June 2018 and April 2019, which were their main anticipated source of revenue.
Less than a year after approval, they filed for bankruptcy as they failed to raise further funding and their main source of revenue did not perform as anticipated.
In light of such examples, it is crucial to mitigate these types of setbacks by having a diversified funding strategy, along with other investors.
Beyond SR&ED, Canadian companies can access various government programs that can help:
Biotech is known for long stagnation periods; companies often spend more than 20 years in development cycles, which is something most founders do not anticipate. Drug and device pipelines can take 7–10 years to bring to market. Without bridge funding or SR&ED claims, funds can dry up during trials or scale-up.
To stay afloat during development lulls or clinical delays, Canadian biotech firms can also explore:
Success is defined differently for different biotech startups. The dream is to create a viable product that achieves commercial success or an airtight exit strategy (acquisition, IPO, or long-term revenue).
Keeping the lights on for five years, or raising large amounts of VC money, or getting a one-hit publication or pitch win, is not the best definition of success, as these are fleeting. Real success comes from a burn rate that aligns with your funding cycles, leveraging available government programs such as SR&ED, and securing non-dilutive funding that supports the entire development cycle.
If your biotech startups are spending money in the development phase, you could be leaving hundreds of thousands of dollars on the table. Before another funding round or another failed pre-clinical trial, get in touch with Leyton to see if you’re eligible for SR&ED and other government programs that can support your development.
Explore our latest insights
More arrow_forward
Harnessing Science for Ecosystem Protection Canada, with its immense ecological diversity, from A...

The Canadian mining sector is transforming. Automation, electrification, and real-time data integ...
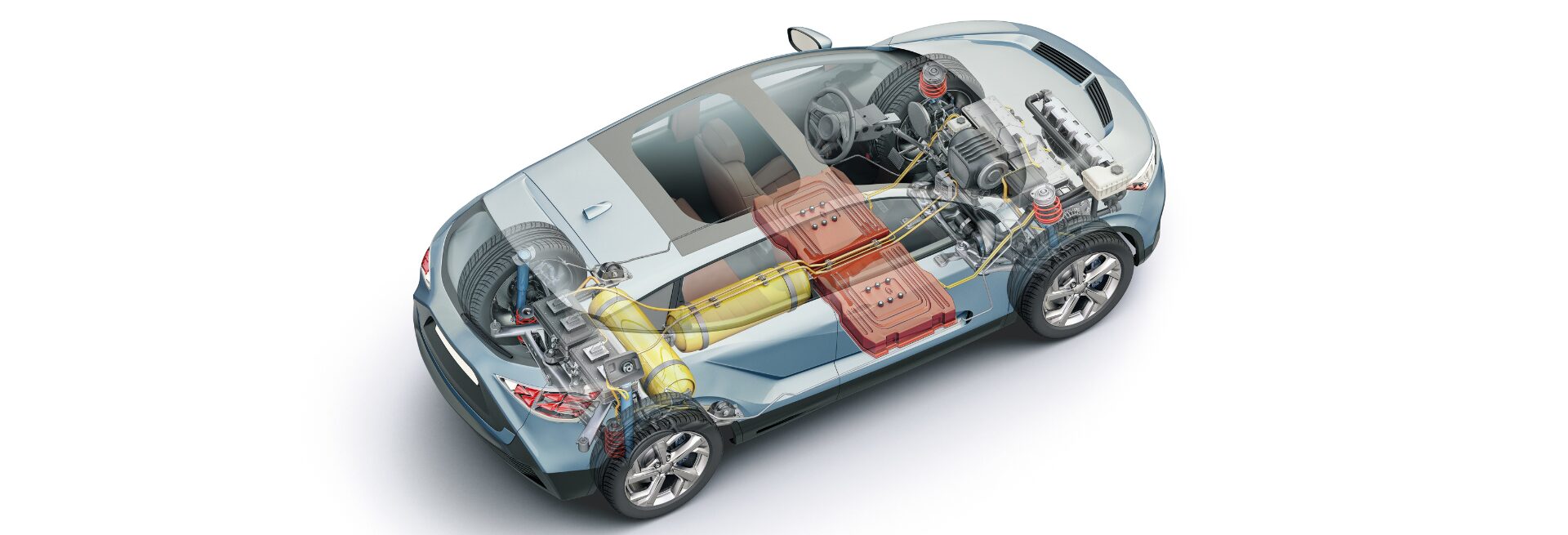
The race toward cleaner, more efficient transportation has accelerated research into lighter, sma...

The Scientific Research & Experimental Development (SR&ED) program is one of Canada’...